Structural Biology and Structure-Based drug discovery services
Our cutting-edge technology platform and extensive experience in structural biology and structure-based drug discovery will significantly accelerate your discovery project.
Your Trusted Partner in Structural Biology & Structure-Based Drug Discovery
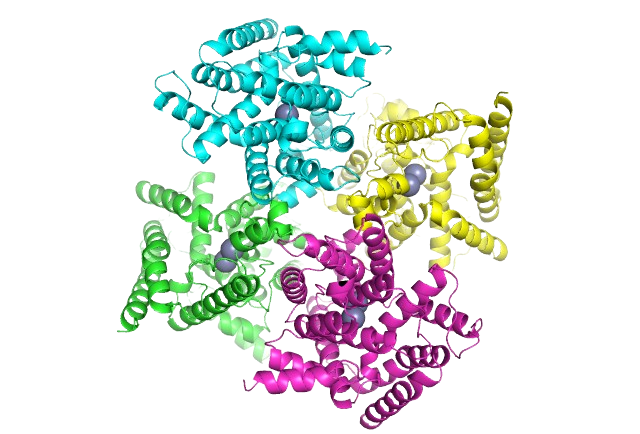
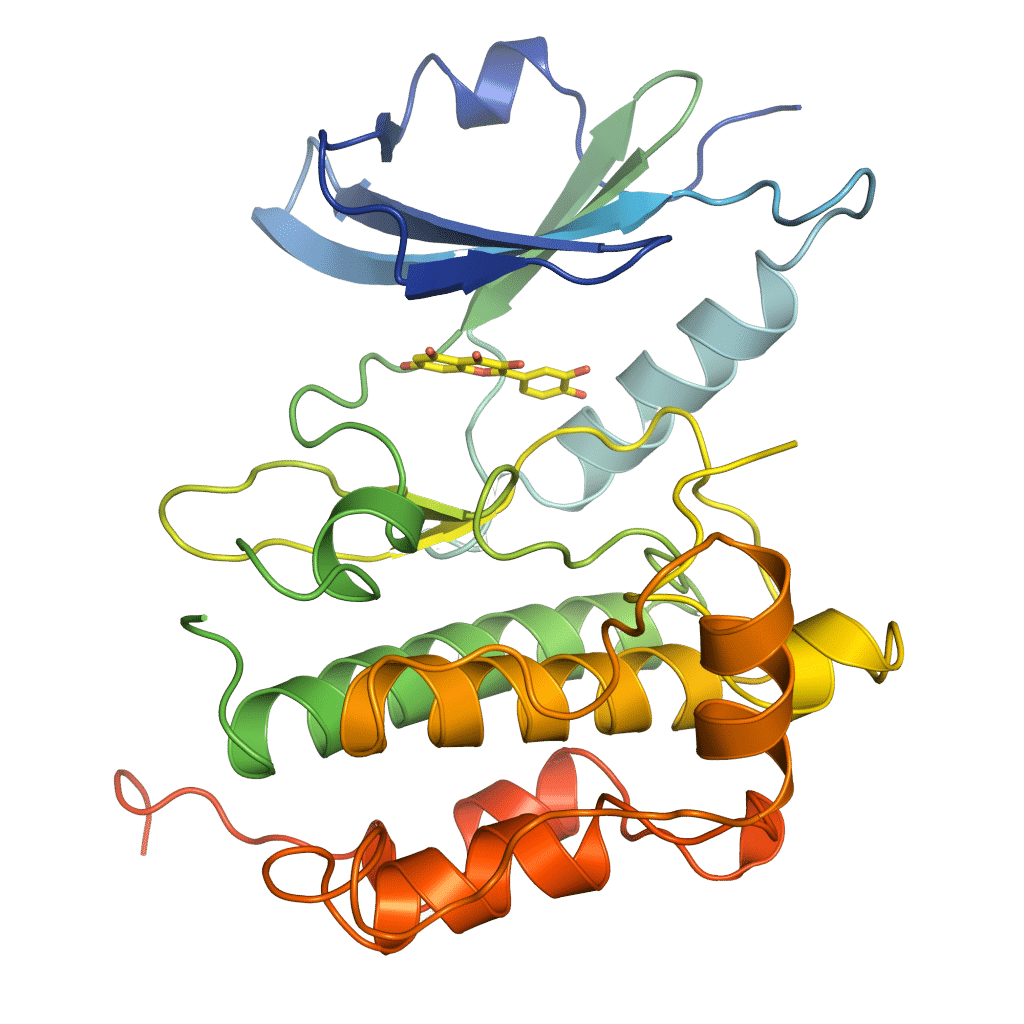
SARomics Biostructures provides structural biology and structure-based drug discovery services to the biotech industry, large pharmaceutical companies, and academic groups. The company was established in 2006 as a research-focused contract research organization (CRO) by scientists with extensive experience in structural biology and structure-based drug design. It rapidly gained recognition as a leading service provider across North and South America, Asia, Europe, Scandinavia, and Australia. Our comprehensive range of outsourcing services is backed by a state-of-the-art technology platform that ensures the success of your project.
An overview of our services is provided below.

MAX IV Laboratory in Lund, home to the BioMax beamline, is one of the world’s best synchrotron radiation facilities, located just a few kilometers from our labs at Medicon Village.
Discover how SARomics Biostructures’ Services can benefit you
Explore our extensive services in structural biology and structure-based drug discovery
Crystallization-Grade Recombinant Proteins For Research And Analytic Purposes
Our comprehensive catalog features high-purity, crystallization-grade drug-target recombinant proteins meticulously expressed and purified to the highest standards in our cutting-edge protein lab. These proteins have undergone rigorous characterization using biophysical methods, and their suitability for crystallization has been unequivocally validated.
Make sure not to miss our featured blog posts!

This article discusses weak chromatography (WAC™), the proprietary fragment screening technique jointly owned by SARomics Biostructures and Red Glead Discovery.

This article discusses some representative projects and company business development activities from 2023.